Portland cement consists primarily of calcium silicates. When mixed with water, a chemical reaction known as hydration creates calcium silicate hydrate (CSH). Needle-like crystals of CSH interlock to hold concrete together, almost like a glue.
Hydration also produces calcium hydroxide, also known as lime. In fact, approximately 15 percent to 25 percent of hydrated portland cement is lime. Lime does not contribute to the strength of concrete and can even be detrimental to a project. For example:
- Lime migrates out of concrete to form efflorescence on the concrete surface.
- When lime leaches out of concrete, it creates voids that increase the porosity of concrete, allowing moisture penetration and corrosion.
- Lime is highly alkaline and contributes to alkali silica reaction (ASR) that can crack concrete.
Pozzolans are supplementary cementitious admixtures that react with lime to form more CSH. The additional CSH increases the cementitious bonds in the concrete to make it stronger. This also makes concrete denser and less porous so concrete is more resistant to penetration by water and chemicals. And because pozzolans consume lime, there is less efflorescence and ASR.
Highly reactive pozzolans like metakaolin outperform other pozzolans and can produce high-performance mixtures with higher strength and lower porosity than could otherwise be achieved.
|
|
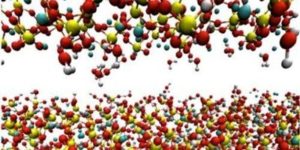
In this simulated microscopic view of cement paste, hydration begins when portland cement particles are surrounded by water. |
|
|
The portland cement begins to dissolve in the water, forming a gel. |
|
|
Crystals begin to form as a result of chemical reactions. |
|
|
Calcium silicate hydrate (CSH) shown here in white, creates interlocking crystals that give concrete its strength. However, as much as 25 percent of the products of hydration become calcium hydroxide (lime), shown in green, and does not contribute to the strength of concrete. Voids are formed by excess water in a mixture. |
|
|
The voids create pores that allow water to penetrate concrete. Since lime is soluble, it dissolves to create even larger pores and allow more liquid and gases to penetrate into the concrete where they can accelerate corrosion. The lime can also be deposited on the concrete surface where it forms efflorescence and disfigures concrete. |
|
|
Inside the concrete, the highly alkaline lime can react with certain types of aggregate to form alkali silica reaction (ASR). ASR expands in the presence of water, creating forces that can crack concrete. |
|
|
As an alternative to ordinary concrete mixtures, high reactivity metakaolin can be used to replace 5 percent to 20 percent of the portland cement in a mixture. The small size of the metakaolin particles fills in the gaps between cement particles to create a denser and creamier cement paste. |
|
|
As a pozzolan, metakaolin reacts with lime formed during cement hydration to form additional CSH. Because there is less lime, the potential for efflorescence and ASR are reduced. |
|
|
The additional CSH formed by metakaolin results in a denser concrete that is stronger and has greater durability due to lower porosity. Plus, the white color of metakaolin brightens the concrete. |
A unique animation explaining how pozzolans improve concrete is available at the Engelhard Web site as part of an online continuing education program.